- Aeviva
- Posts
- Biological Drugs and Gene Therapy: What They Are, How They Work, and Why They're Revolutionary
Biological Drugs and Gene Therapy: What They Are, How They Work, and Why They're Revolutionary
Traditional drugs are simple chemicals made in labs, but biologics are living proteins made by cells, and gene therapy rewrites your DNA to cure diseases

Estimated Read Time: 6 minutes
⚠️ Note: This newsletter covers complex biotechnology topics that may be more technical than usual, but we've explained everything in the simplest terms possible.
Traditional drugs like aspirin or antibiotics are simple chemicals synthesized in factories.
Biological drugs like insulin (Humalog, Lantus), cancer immunotherapies (Keytruda, Opdivo), and arthritis treatments (Humira, Enbrel) are complex proteins manufactured inside living cells.
Gene therapy goes further, inserting new genes into your cells to permanently fix genetic diseases like sickle cell anemia and inherited blindness.
Today's Issue
Main Topic: Understanding biological drugs and gene therapy - what they are, how they're made, and how they work differently from traditional medicine
Subtitles:
What biological drugs are: proteins, antibodies, and living molecules vs chemical drugs
Examples: insulin, monoclonal antibodies, cancer immunotherapy, and biologics for autoimmune diseases
How biologics are manufactured inside living cells (bacteria, yeast, mammalian cells)
What gene therapy is: inserting, deleting, or editing genes to cure disease
How gene therapy works: viral vectors, CRISPR, and permanent genetic fixes
Current gene therapy successes: treating blindness, sickle cell disease, and cancer
Abstract: Biological drugs (biologics) are large complex molecules (typically 1000-25,000+ atoms) including proteins, antibodies, and nucleic acids produced inside living cells through recombinant DNA technology, contrasting with traditional small-molecule drugs (100-1000 atoms) chemically synthesized in factories. Major biologic classes include recombinant proteins like insulin (Humalog, Lantus, Novolog) produced in E. coli or yeast replacing animal-derived insulin, monoclonal antibodies (mAbs) like adalimumab/Humira ($20 billion annually treating rheumatoid arthritis, Crohn's disease) targeting specific immune proteins, checkpoint inhibitors like pembrolizumab/Keytruda and nivolumab/Opdivo blocking PD-1 receptors enabling immune system cancer destruction, and fusion proteins like etanercept/Enbrel blocking TNF-alpha in autoimmune diseases. Manufacturing requires inserting human genes into bacteria, yeast, or mammalian cell lines (CHO cells most common) that produce desired proteins, followed by complex purification costing $100-300 million per manufacturing facility. Gene therapy delivers functional genes into patient cells using viral vectors (modified viruses carrying therapeutic genes) like AAV (adeno-associated virus) for eye diseases and hemophilia, lentiviruses for ex vivo therapies where cells are removed, modified, and returned, or CRISPR-Cas9 systems directly editing DNA to fix mutations. Approved gene therapies include Luxturna (AAV-delivered RPE65 gene treating inherited blindness, $850,000 one-time treatment), Zolgensma (AAV-delivered SMN1 gene treating spinal muscular atrophy, $2.1 million), Casgevy (CRISPR editing for sickle cell disease and beta-thalassemia, approved 2023), and CAR-T therapies (Kymriah, Yescarta) genetically modifying immune cells to attack blood cancers.
The pharmaceutical revolution over the past 30 years has shifted from discovering small chemical molecules that modify biological processes to creating biological molecules that replicate or enhance the body's own systems, and most recently to directly rewriting genetic code to cure diseases at their root cause. Traditional drugs like aspirin (21 atoms), penicillin (41 atoms), or statins (40-60 atoms) are relatively simple chemical structures that can be synthesized through chemical reactions in factories, stored at room temperature for years, and taken as pills because they survive digestion.
Biological drugs are fundamentally different: they're massive protein molecules (insulin has 787 atoms, monoclonal antibodies have 25,000+ atoms) produced inside living organisms (bacteria, yeast, or mammalian cells engineered to manufacture human proteins), requiring refrigeration because they degrade quickly, and administered by injection because stomach acid would destroy them. Gene therapy represents the next evolution, moving beyond delivering proteins to delivering the genetic instructions for cells to make their own proteins permanently.
Understanding how biologics are manufactured (inserting human genes into bacteria or cell lines, purifying proteins, maintaining sterility), what types exist (recombinant proteins replacing missing enzymes or hormones, monoclonal antibodies targeting specific disease proteins, fusion proteins blocking inflammatory signals), and how gene therapy works (using modified viruses as delivery vehicles to insert genes into specific cells, or using CRISPR to directly edit DNA sequences) reveals why these treatments are revolutionary for previously incurable diseases but cost $50,000-$2,000,000 per patient annually or per treatment.
This newsletter examines what makes biologics different from traditional drugs, how major biologic classes work with specific examples, how gene therapy delivers and integrates new genetic material, and what diseases are currently being treated or cured through these technologies.
Are You Ready to Climatize?
Climatize is an investment platform focused on renewable energy projects across America.
You can explore vetted clean energy offerings, with past projects including solar on farms in Tennessee, grid-scale battery storage in New York, and EV chargers in California.
Each project is reviewed for transparency and offers people access to fund development and construction loans for renewable energy in local communities.
As of November 2025, more than $13.2 million has been invested through the platform across 28 renewable energy projects. To date, over $3.6 million has already been returned to our growing investor base. Returns are not guaranteed, and past performance does not predict future results.
Check out Climatize to explore clean energy projects raising capital. Minimum investments start with as little as $10.
Climatize is an SEC-registered & FINRA member funding portal. Crowdfunding carries risk, including loss.
1. What Biological Drugs Are: Proteins, Antibodies, and Living Molecules vs Chemical Drugs 🧬💊
Traditional small-molecule drugs:
Aspirin (180 molecular weight), penicillin (334 MW), ibuprofen (206 MW), statins (400-500 MW). These are simple chemical structures with 15-100 atoms, synthesized through chemical reactions in factories.
They're stable at room temperature, survive stomach acid, and can be taken as pills.
Biological drugs (biologics):
Massive complex molecules (15,000-150,000 molecular weight) including proteins, antibodies, and nucleic acids.
Insulin has 5,808 MW (787 atoms), monoclonal antibodies have 150,000 MW (25,000+ atoms). These cannot be chemically synthesized - they must be produced inside living cells.
Key differences:
Feature | Chemical Drugs | Biological Drugs |
|---|---|---|
Size | Small (100-1000 atoms) | Large (1000-25,000+ atoms) |
Structure | Simple, defined | Complex, 3D protein structures |
Manufacturing | Chemical synthesis in factories | Living cells (bacteria, yeast, mammalian) |
Stability | Stable, room temperature storage | Fragile, requires refrigeration |
Administration | Pills (survive digestion) | Injection (destroyed by stomach acid) |
Cost | $1-100 per dose | $1,000-50,000+ per dose |

Major classes:
Recombinant proteins: Human proteins produced in bacteria or cells. Examples: insulin, growth hormone, clotting factors for hemophilia.
Monoclonal antibodies (mAbs): Antibodies targeting specific proteins involved in disease. Examples: Humira, Keytruda, Opdivo. Identified by "-mab" suffix.
Fusion proteins: Engineered proteins combining parts of different proteins. Examples: Enbrel (TNF receptor fused to antibody).
💡 Fun Fact: The first biological drug was recombinant human insulin (Humulin) approved in 1982. Before this, diabetics used insulin extracted from pig or cow pancreases, which caused allergic reactions. Humulin was produced by inserting the human insulin gene into E. coli bacteria.
2. Examples: Insulin, Monoclonal Antibodies, Cancer Immunotherapy, and Biologics for Autoimmune Diseases 💉🎯
Recombinant insulin:
Drugs: Humalog (lispro), Novolog (aspart), Lantus (glargine), Levemir (detemir), Tresiba (degludec).

Novo Nordisk NovoRapid insulin
How it works: Type 1 diabetics don't produce insulin (hormone regulating blood sugar). Recombinant insulin replaces missing hormone, allowing cells to absorb glucose from blood.
Types: Rapid-acting (works in 15 minutes, lasts 3-5 hours, taken before meals), long-acting (works in 1-2 hours, lasts 12-24 hours, taken once or twice daily).
Manufacturing: Human insulin gene inserted into E. coli bacteria or yeast. Bacteria produce human insulin protein, which is purified and formulated.
Cost: $300-700 per vial in US. Manufacturing cost is $5-10 per vial, the rest is profit and distribution.
Monoclonal antibodies for autoimmune diseases:
Humira (adalimumab): $20 billion annual sales before biosimilars. Treats rheumatoid arthritis, Crohn's disease, ulcerative colitis, psoriasis. Monoclonal antibody targeting TNF-alpha (tumor necrosis factor), preventing it from activating inflammatory pathways. Dose: 40mg injection every 2 weeks. Cost: $6,000-7,000 monthly before biosimilars.
Enbrel (etanercept): Treats rheumatoid arthritis, psoriasis. Fusion protein combining TNF receptor with antibody, acting as "decoy" binding TNF-alpha before it reaches real receptors. Dose: 50mg injection weekly. Cost: $5,000-6,000 monthly.

Remicade (infliximab): Treats Crohn's disease, ulcerative colitis, rheumatoid arthritis. Monoclonal antibody targeting TNF-alpha. Dose: IV infusion every 4-8 weeks. Cost: $4,000-8,000 per infusion.
Cancer immunotherapy (checkpoint inhibitors):
Keytruda (pembrolizumab): $25 billion annual sales. Treats melanoma, lung cancer, head and neck cancer, bladder cancer, 20+ cancer types. Monoclonal antibody blocking PD-1 receptor on immune T cells.
Dose: 200mg IV infusion every 3 weeks. Response rate: 30-45% of PD-L1 positive tumors. Cost: $150,000-200,000 yearly.
Opdivo (nivolumab): Treats melanoma, lung cancer, kidney cancer, liver cancer. Similar mechanism blocking PD-1 receptor. Cost: $150,000-200,000 yearly.
💡 Critical Context: Checkpoint inhibitors work by unleashing the patient's own immune system, explaining both success (durable remissions, sometimes cures) and side effects (autoimmune reactions attacking normal tissues).
3. How Biologics Are Manufactured Inside Living Cells 🏭🦠
Why living cells are necessary:
Proteins are chains of amino acids (insulin has 51 amino acids, antibodies have 1300+ amino acids) that must fold into precise 3D shapes. Only living cells have the molecular machinery (ribosomes, chaperones, post-translational modification systems) to properly make and fold proteins.
Manufacturing process:
Step 1: Gene insertion
Scientists identify the gene for the desired protein. The gene is inserted into a plasmid (circular DNA) or directly into the host cell's DNA.
Step 2: Host cell selection
E. coli bacteria: Fast-growing, easy to work with, cheap. Used for simple proteins like insulin, growth hormone. Cannot perform complex modifications like adding sugars to proteins (glycosylation).
Yeast: Can perform some modifications bacteria can't. Used for vaccines, some enzymes.
Mammalian cells (CHO - Chinese hamster ovary cells): Can perform all human protein modifications. Required for complex proteins like antibodies. Slower and more expensive but necessary for most biologics.
Step 3: Cell culture (fermentation)
Host cells are grown in massive bioreactors (10,000-25,000 liters) containing nutrients. Cells multiply and produce the desired protein, which either accumulates inside cells or is secreted into culture medium. Fermentation takes 10-21 days.
Step 4: Harvesting and purification
Cells are removed (centrifugation or filtration). Protein is purified using multiple steps: chromatography (separating by size, charge, binding properties), filtration, viral inactivation. Purification is the most expensive part, requiring 5-10 steps to achieve 99.9%+ purity.
Step 5: Formulation
Purified protein is formulated with stabilizers, preservatives, pH buffers, and filled into vials or pre-filled syringes.
Step 6: Quality control
Extensive testing for purity, potency, sterility, endotoxins, and correct protein structure. Batch testing takes 2-6 months before release.

Antibody bioreactor
Why biologics are expensive:
Manufacturing facility costs $100-300 million to build. Production takes 6-9 months from start to finished product (chemical drug synthesis takes days to weeks). Complex purification and quality control. Refrigerated distribution. Patents last 12-20 years, allowing high prices before biosimilar competition.
💡 Pro Tip: When you see "-mab" at the end of a drug name (adalimumab, pembrolizumab, nivolumab), it's a monoclonal antibody. "-cept" indicates a receptor fusion protein (etanercept). "-ase" indicates an enzyme.
4. What Gene Therapy Is: Inserting, Deleting, or Editing Genes to Cure Disease 🧬✂️
Gene therapy delivers functional genes into patient cells to replace defective genes, silence disease-causing genes, or add new capabilities to cells.
The concept:
Many diseases are caused by single-gene mutations. Sickle cell disease (mutation in beta-globin gene), hemophilia (mutation in clotting factor genes), inherited blindness (mutations in retinal genes), spinal muscular atrophy (missing SMN1 gene).
If you could insert a working copy of the gene, you could cure the disease permanently.
Three main approaches:
Gene addition: Add a functional copy of missing or defective gene without removing the bad gene. The good gene compensates.
Gene silencing: to block expression of disease-causing genes.
Gene editing (CRISPR): Directly cut out and replace defective DNA sequences with correct ones, permanently fixing the mutation.
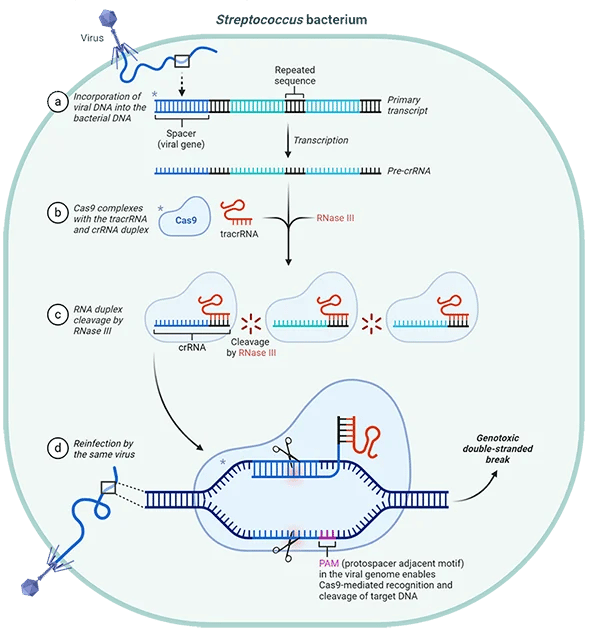
CRISPR Cas-9 technology
Delivery systems (vectors):
Viral vectors (most common): Modified viruses stripped of disease-causing genes, carrying therapeutic genes instead. Viruses naturally evolved to efficiently deliver DNA/RNA into cells.
AAV (adeno-associated virus): Small virus infecting many cell types without causing disease. Used for eye diseases, hemophilia, spinal muscular atrophy. Delivers genes that stay active for years but don't integrate into chromosomes.
Lentivirus (modified HIV): Integrates into chromosomes, providing permanent gene expression. Used for ex vivo therapies (cells removed, modified, returned) like CAR-T and sickle cell treatments.
Non-viral delivery: Lipid nanoparticles (used in mRNA vaccines), electroporation (using electric pulses), direct DNA injection. Less efficient than viruses but avoids immune responses.
5. How Gene Therapy Works: Viral Vectors, CRISPR, and Permanent Genetic Fixes 🦠✂️💉
In vivo gene therapy (genes delivered directly into patient):
Example: Luxturna for inherited blindness

Disease: retinal dystrophy (mutation in RPE65 gene causes progressive blindness starting in childhood).
Mechanism: AAV infects retinal cells, delivering working RPE65 gene.
Cells start producing RPE65 protein (enzyme converting vitamin A derivatives needed for vision).
Vision improves within weeks and is maintained for years (longest follow-up is 10+ years).
Cost: $425,000 per eye ($850,000 total).
Results: Most patients gain ability to navigate in low light, some gain significant vision improvement.
Example: Zolgensma for spinal muscular atrophy (SMA)
Disease: Missing or defective SMN1 gene causes motor neuron death, leading to muscle weakness, respiratory failure, and death by age 2 in severe cases.
Treatment: One-time IV infusion of AAV9 vector carrying SMN1 gene.
Mechanism: AAV9 crosses blood-brain barrier, infecting motor neurons in spinal cord. Neurons produce SMN protein, preventing death and restoring function. Treatment must be given before age 2 for best results.
Cost: $2.1 million.
Results: Babies treated before symptoms develop can walk, talk, and live normal lives.
Ex vivo gene therapy (cells removed, modified, and returned):
Example: CAR-T therapy for blood cancers
Diseases: B-cell lymphomas, B-cell leukemias that didn't respond to chemotherapy.
Drugs: Kymriah (tisagenlecleucel), Yescarta (axicabtagene ciloleucel).
Process: Patient's T cells are removed, sent to manufacturing facility where lentivirus delivers CAR (chimeric antigen receptor) gene. CAR is an engineered protein combining antibody portion (recognizing CD19 on cancer cells) with T cell activation domain. Modified T cells are expanded to billions over 2-4 weeks. Patient receives chemotherapy to eliminate existing immune cells. CAR-T cells are infused back.
Mechanism: CAR-T cells recognize and kill any cell displaying CD19 (cancer cells and normal B cells). Patient's immune system is rebuilt with CAR-T cells that patrol for cancer forever.
Results: 40-50% achieve complete remission. Many remain cancer-free for 5+ years (possible cure). Side effects can be severe (cytokine release syndrome).
Cost: $373,000-475,000.
CRISPR gene editing:
Example: Casgevy for sickle cell disease and beta-thalassemia

Diseases: Sickle cell disease (mutation causing abnormal hemoglobin, painful crises), beta-thalassemia (reduced beta-globin production, severe anemia).
Treatment (approved December 2023): Patient's blood stem cells are collected. CRISPR-Cas9 edits BCL11A gene (which normally suppresses fetal hemoglobin production). Editing causes stem cells to produce fetal hemoglobin (which doesn't sickle and works normally) even in adults. Patient receives chemotherapy destroying existing bone marrow. Edited stem cells are infused, repopulating bone marrow.
Mechanism: By reactivating fetal hemoglobin, treatment compensates for defective adult hemoglobin. CRISPR makes a precise DNA cut, cell's repair mechanisms introduce small errors that disable the suppressor gene.
Results: 28 of 29 sickle cell patients in trials were crisis-free for 1+ years. All beta-thalassemia patients became transfusion-free.
Cost: Estimated $2-3 million.
💡 Critical Context: Gene therapy works best for single-gene diseases affecting accessible tissues (blood cells, eyes, liver, muscles). Complex diseases involving multiple genes (most cancers, Alzheimer's, diabetes) can't be cured by adding or fixing one gene.
6. Current Gene Therapy Successes: Treating Blindness, Sickle Cell Disease, and Cancer 🏆🎯
Approved gene therapies (as of 2024):
Luxturna: Inherited retinal dystrophy, $850,000, approved 2017.
Zolgensma: Spinal muscular atrophy, $2.1 million, approved 2019.
Kymriah: B-cell lymphoma and leukemia, $475,000, approved 2017.
Yescarta: B-cell lymphoma, $373,000, approved 2017.
Breyanzi: B-cell lymphoma, $410,000, approved 2021.
Abecma: Multiple myeloma, $419,000, approved 2021.
Hemgenix: Hemophilia B, $3.5 million (most expensive drug), approved 2022.
Roctavian: Hemophilia A, $2.9 million, approved 2023.
Casgevy: Sickle cell disease and beta-thalassemia, $2-3 million estimated, approved December 2023 (first CRISPR therapy).
Lyfgenia: Sickle cell disease, approved December 2023.
💡 Pro Tip: Gene therapy costs seem astronomical ($2-3 million), but compare to lifetime costs. Hemophilia clotting factor costs $300,000-500,000 annually for life (potentially $20-30 million over lifetime). One-time curative gene therapy could be cost-effective despite high upfront price.
The Proven System Fitness Instructors Use to Grow Online
Discover Kajabi’s 30 Days to Launch: Scale Your Fitness Business Online guide. Launch online programs, memberships, and digital products while keeping in-person clients. Step-by-step roadmap, success stories, and AI prompts included.
Takeaways
Biological drugs are large complex proteins (insulin 787 atoms, monoclonal antibodies 25,000+ atoms) manufactured inside living cells (bacteria, yeast, mammalian CHO cells) rather than chemically synthesized, including recombinant proteins like insulin (Humalog, Lantus) replacing missing hormones, monoclonal antibodies like Humira ($20 billion annually) targeting TNF-alpha for autoimmune diseases, and checkpoint inhibitors like Keytruda and Opdivo blocking PD-1 receptors enabling immune system cancer destruction.
Manufacturing biologics requires inserting human genes into host cells, growing in massive bioreactors (10,000-25,000 liters), and complex multi-step purification taking 6-9 months, costing $100-300 million for facilities and $1,000-50,000 per dose for final product, with refrigeration required throughout distribution because proteins degrade quickly unlike stable chemical drugs stored at room temperature.
Gene therapy delivers functional genes using viral vectors (AAV for eye diseases like Luxturna $850,000, lentivirus for ex vivo CAR-T therapies $373,000-475,000) or CRISPR editing (Casgevy for sickle cell disease $2-3 million approved December 2023), providing one-time potentially curative treatments by permanently fixing genetic defects, with 10+ approved therapies treating inherited blindness, spinal muscular atrophy, hemophilia, blood cancers, and now sickle cell disease.
Feedback & Sponsorship
What'd you think of this week's newsletter? Hit reply to let us know. Did we crush it? Blow your mind? We read every response.
Want your brand in front of hundreds of thousands of readers? Contact us for sponsorship opportunities [email protected]
Want more where that came from? Head to our website
Save up to $100 on Qi35 This Holiday Season
The holiday season is here and TaylorMade is making it ridiculously easy to score something everyone on your list will actually love. Right now you can save $100 on Qi35 drivers and $50 on Qi35 fairway woods and rescue clubs. This is the kind of gift that pays off round after round.




Reply